Unique Access provides access to an extensive treatment protocol for Spinal Muscular Atrophy (SMA) type 2 & 3, which utilises only the best stem cells, immune boost, diet, extensive rehabilitation, and many supportive therapies and supplements. This effective combination of the most advanced medical technologies with systematic physical and occupational therapies has helped previously treated patients achieve significant improvements.
Why Stem Cells Work For Spinal Muscular Atrophy (SMA)
Stem Cell Therapy is considered to be an alternative method for treating Spinal Muscular Atrophy (SMA) and other neurodegenerative disorders. Stem cell transplantation is a potential therapeutic strategy not only via cell replacement but also by modification of the extracellular motor neuronal environment, through a trophic and neuroprotective effect. A variety of cell sources have been considered for cell therapy.
Many of the treated patients showed major improvements in motor function, muscle tone and strength, speech and swallowing, balance and coordination, and fine and gross motor skills. Moreover, the disease progression has been reported to be significantly slower after stem cell transplantation.
Mechanisms
Mesenchymal Stem Cells (MSCs) are very attractive multipotent stem cells for Spinal Muscular Atrophy (SMA) cell therapy because of their great plasticity and their ability to provide the host tissue with growth factors and to modulate the host immune system.
The MSCs transplantation increases neuron survival and prevents gliosis, as microglia and astrocytes are both the target and cause of neuroinflammation, MSCs can rescue neurons and oligodendrocytes from apoptosis through the release of trophic and anti-apoptotic molecules, resulting in the induction of a neuroprotective microenvironment.
In addition, Mesenchymal Stem Cells can promote the proliferation and maturation of local neural precursor cells, leading to their differentiation into mature neurons and oligodendrocytes.
Improvements
Spinal Muscular Atrophy (SMA) patients treated with stem cells usually observe improvements in the following areas:
- Motor Function
- Balance
- Muscle tone and strength
- Coordination
- Neuropathic Pain
- Fatigue
- Tremors
- Speech
- Fine and gross motor
- Swallowing
- Slowed down progression and more
Our Promise
We believe that there is always hope and that patients deserve access to effective and safe treatments. We are independent with an in-house medical department. We combine internationally accredited hospitals, next generation treatments, unique products and services that are integrative and effective to ensure best possible treatment results.
Stem Cells
In terms of stem cells we will make sure that the patient will receive the correct and necessary stem cell type, quality, quantity and viability. Our exclusive research partner guarantee a stem cell viability of 95%, many injections have a staggering viability of 98-99%.
Supportive Therapies & Remedies
- Hyperbaric Oxygen Chamber (HBOT)
- Hemo Oxygen Therapy (HOT)
- IV Vitamin Drips
- Immune-Boosting Supplements (e.g. GcMAF)
Partner Hospital
The treatment will take place in an internationally accredited tertiary care hospital and not in a hotel or clinic. This is important for the patient’s safety and care as the patient will have access to all specialized departments & specialist doctors which will further increase the treatments efficiency.
What is Spinal Muscular Atrophy (SMA) ?
Spinal Muscular Atrophy (SMA) is a hereditary disease affecting the motor neurons in the spinal cord.
Motor neurons control the voluntary muscle movements in the body, thus, Spinal Muscular Atrophy (SMA) results in significant muscle degeneration and weakness, affected muscle tone, and affected balance and coordination.
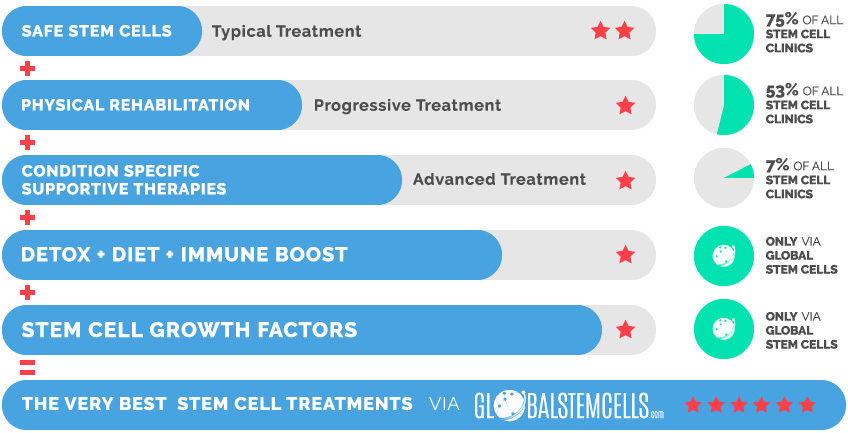
The Very Best Stem Cell Treatments via globalstemcells.com
- Dubowitz, V. (2009).
“Ramblings in the history of spinal muscular atrophy”. Neuromuscular Disorders. 19 (1): 69–73. doi: 10.1016/j.nmd.2008.10.004. PMID 18951794.
- Sugarman, E. A.; Nagan, N.; Zhu, H.; Akmaev, V. R.; Zhou, Z.; Rohlfs, E. M.; Flynn, K.; Hendrickson, B. C.; Scholl, T.; Sirko-Osadsa, D. A.; Allitto, B. A. (2011).
“Pan-ethnic carrier screening and prenatal diagnosis for spinal muscular atrophy: Clinical laboratory analysis of >72 400 specimens”. European Journal of Human Genetics. 20 (1): 27–32. doi: 10.1038/ejhg.2011.134. PMC 3234503. PMID 21811307.
- Little, S. E.; Janakiraman, V.; Kaimal, A.; Musci, T.; Ecker, J.; Caughey, A. B. (2010).
“The cost-effectiveness of prenatal screening for spinal muscular atrophy”. American Journal of Obstetrics and Gynecology. 202 (3): 253.2e1. doi: 10.1016/j.ajog.2010.01.032. PMID 20207244.
- Prior, T. W.; Professional Practice Guidelines Committee (2008).
“Carrier screening for spinal muscular atrophy”. Genetics in Medicine. 10 (11): 840–842. doi: 10.1097/GIM.0b013e318188d069. PMC 3110347. PMID 18941424.
- Bach, J. R.; Niranjan, V.; Weaver, B. (2000).
“Spinal Muscular Atrophy Type 1: A Noninvasive Respiratory Management Approach”. Chest. 117 (4): 1100–1105. doi: 10.1378/chest.117.4.1100. PMID 10767247.
- Chen, Y. S.; Shih, H. H.; Chen, T. H.; Kuo, C. H.; Jong, Y. J. (2011).
“Prevalence and Risk Factors for Feeding and Swallowing Difficulties in Spinal Muscular Atrophy Types II and III”. The Journal of Pediatrics. 160 (3): 447–451.e1. doi: 10.1016/j.jpeds.2011.08.016. PMID 21924737.
- Shababi, M.; Habibi, J.; Yang, H. T.; Vale, S. M.; Sewell, W. A.; Lorson, C. L. (2010).
“Cardiac defects contribute to the pathology of spinal muscular atrophy models”. Human Molecular Genetics. 19 (20): 4059–4071. doi: 10.1093/hmg/ddq329. PMID 20696672.
- Bevan, A. K.; Hutchinson, K. R.; Foust, K. D.; Braun, L.; McGovern, V. L.; Schmelzer, L.; Ward, J. G.; Petruska, J. C.; Lucchesi, P. A.; Burghes, A. H. M.; Kaspar, B. K. (2010).
“Early heart failure in the SMNΔ7 model of spinal muscular atrophy and correction by postnatal scAAV9-SMN delivery”. Human Molecular Genetics. 19 (20): 3895–3905. doi: 10.1093/hmg/ddq300. PMC 2947399. PMID 20639395.
- Von Gontard, A.; Zerres, K.; Backes, M.; Laufersweiler-Plass, C.; Wendland, C.; Melchers, P.; Lehmkuhl, G.; Rudnik-Schöneborn, S. (2002).
“Intelligence and cognitive function in children and adolescents with spinal muscular atrophy”. Neuromuscular Disorders. 12 (2): 130–136. Neuropsychol Rev. 18 (4): 339–66. doi: 10.1016/S0960-8966(01)00274-7. PMID 11738354.
- Zhou, Haiyan; Meng, Jinhong; Marrosu, Elena; Janghra, Narinder; Morgan, Jennifer; Muntoni, Francesco (2015).
“Repeated low doses of morpholino antisense oligomer: An intermediate mouse model of spinal muscular atrophy to explore the window of therapeutic response”. Human Molecular Genetics. 24 (22): 6265–77; 6265. doi: 10.1093/hmg/ddv329. PMC 4614699. PMID 26264577.


 English
English